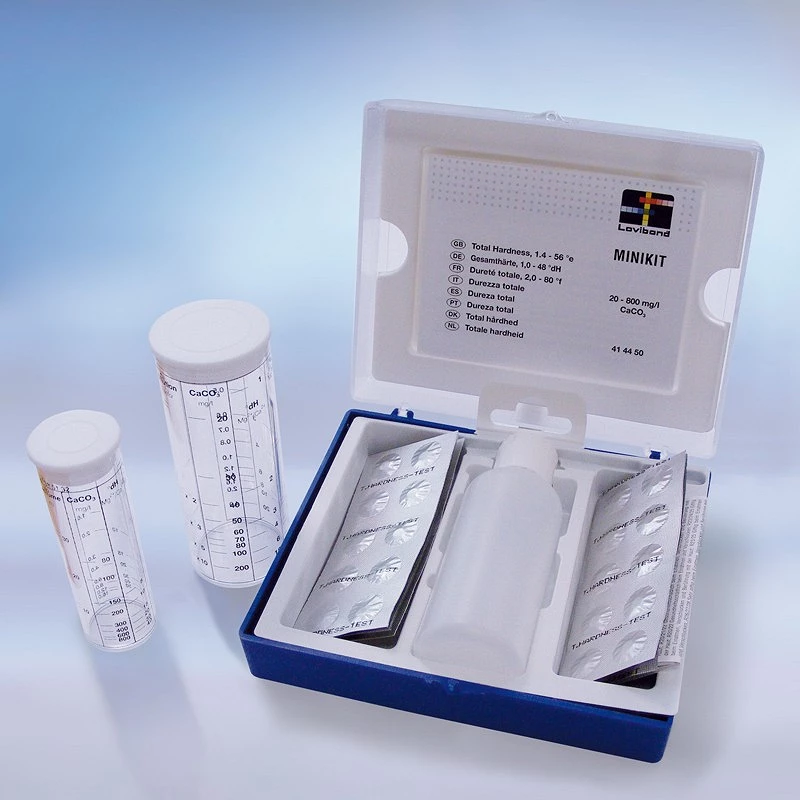
MiniKit for calcium hardness analysis

- Minikit calcium hardness analysis
- With a scale of 20-800 mg/l CaCO3.
- Professional case
- If the hardness is higher than 120, it contains a high content of minerals such as magnesium and calcium salts
Bought together Usually
 Actual product
Actual product
Technical characteristics
Calcium Hardness Analysis
- Minikit calcium hardness analysis
- With a scale of 20-800 mg/l CaCO3.
- Professional case
- If the hardness is higher than 120, it contains a high content of minerals such as magnesium and calcium salts
Hard water.
In chemistry, hard water is water with a hardness of more than 120 mg CaCO3/l.
This means that it contains a high level of minerals, in particular magnesium and calcium salts.
These are the cause of water hardness, and the degree of hardness is directly proportional to the concentration of metallic salts.
Hard water can be made soft again by adding sodium or potassium carbonate to precipitate it as carbonate salts, or by ion exchange with brine in the presence of zeolite or synthetic resins.
what is hard water?
Older generations have coined the phrase "hard water" because it makes cleaning difficult. The difficulty in cleaning is due to the calcium and magnesium compounds it contains. Freshwater sources usually contain varying amounts of calcium and magnesium. Water dissolves, suspends, or exchanges trace compounds and elements from the many things it comes into contact with.
Total water hardness is the measure of the total concentration of calcium and magnesium, the two most frequent bivalent metal ions, although in some geographic locations iron, aluminium and magnesium are also present at elevated levels.
Types of hardness.
Water hardness has a shared distinction between temporary (or carbonate) hardness and permanent (or non-carbonate) hardness.
Temporary hardness is produced by carbonates and can be removed by boiling water or by the addition of lime (calcium oxide). While permanent hardness cannot be removed by boiling water, it is usually caused by the presence of calcium and magnesium sulphate or chlorides in the water, which become more soluble as the temperature rises.
Health problems.
Some studies have shown that there is a weak inverse relationship between water hardness and cardiovascular disease in men above the level of 170 mg of calcium carbonate per litre in water. The World Health Organisation has reviewed the evidence and concluded that the data were inadequate to allow a recommendation for a hardness level.
Data Sheet
- Type of case
- Manual
Verified Reviews
Might Also like